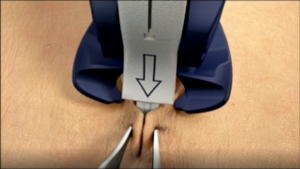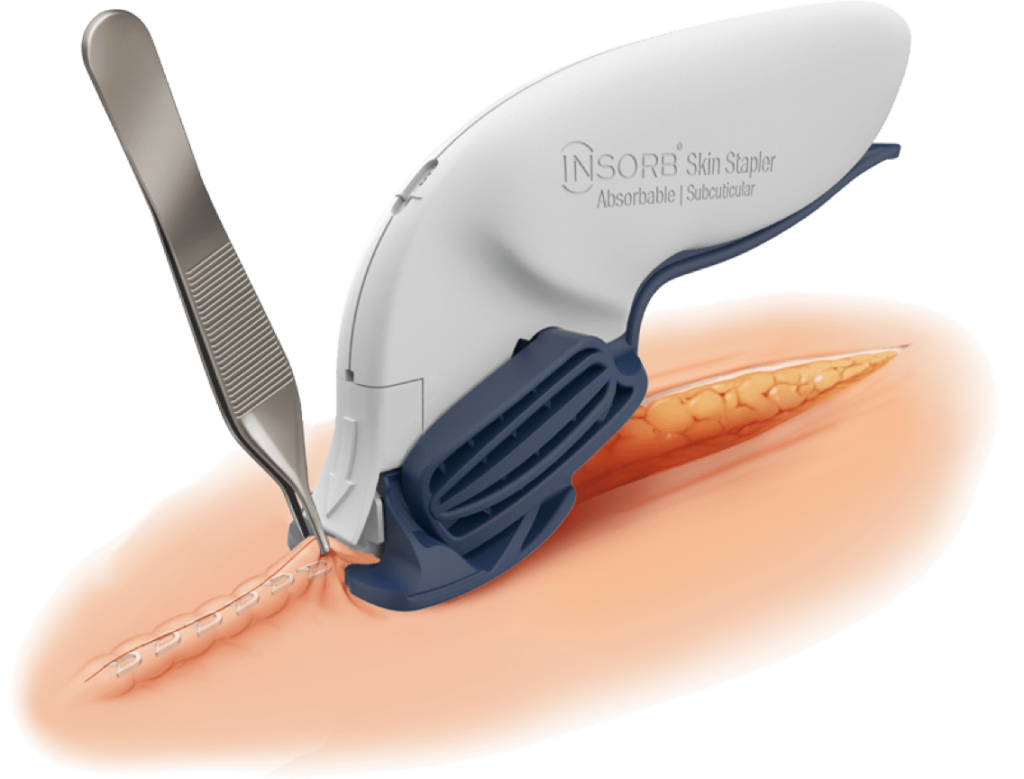

The INSORB Skin Stapler
The INSORB Skin Stapler is a subcuticular, interrupted closure modality that is designed to create uniform, symmetric dermis-to-dermis closures.

The Staple
The INSORB Absorbable Skin Staple
is composed of a co-polymer derived
from polylactide-polyglycolide with a
well-established¹ history in wound closure.

INSORB Stapler
The patented INSORB Skin Stapler is
a sterile, single patient use device that
deploys 30 absorbable staples, sufficient
to close an incision up to 21 cm in length.

Blue Triangles
Keep the blue triangles fully covered by
the tissue. This will assist with proper
alignment of the staples and minimize
external placements.
Why INSORB?
Benefits of INSORB
vs. Suture
The INSORB skin stapler is designed to create uniform, symmetric dermis-to-dermis closures.
- Increases skin eversion, which may improve healing and cosmesis
- May reduce operative time²
- May result in reduced hand fatigue
- Eliminates risk of needlestick injuries at the time of closure
Benefits of INSORB
vs. Metal Staples
The INSORB skin stapler eliminates the percutaneous insult created by metal skin staples.
- Eliminates the pain and discomfort of percutaneous metal skin staples and potential “railroad track” scars
- Evidence of reduced wound complications vs. metal staples³
- Eliminates cost, patient inconvenience, anxiety and discomfort associated with percutaneous metal staple removal
INSORB Staples vs. Metal Staples
There are multiple factors that can affect patient results and recovery and these results may not be representative.
¹ Gunatillake P.A. and A. R. (2003). Biodegradable Synthetic Polymer for Tissue Engineering. European Cells and Materials, CSIRO Molecular Science. Volume 5.
² Madsen, Annetta Madeline, et al. Absorbable Subcuticular Staples Compared with Suture for Cesarean Closure. Obstet Gynecol. 2015; vol. 125.
³ Schrufer-Poland, T. L., Ruiz, M. P., Kassar, S., Tomassian, C., Algren, S. D., & Yeast, J. D. (2016). Incidence of wound complications in cesarean deliveries following closure with absorbable subcuticular staples versus conventional skin closure techniques. European Journal of Obstetrics & Gynecology and Reproductive Biology, 206, 53-56. doi:10.1016/j.ejogrb.2016.07.501